Sudden Cardiac Death in Athletes — Lessons From My Reporting
The sudden death of a teammate is what got me into journalism in the first place
You have probably read by now that Buffalo Bills safety Damar Hamlin suffered a cardiac arrest during a game on January 2. I was delighted to read, a few days later, that he was awake and recovering.
I don’t know what caused Hamlin’s cardiac arrest, and don’t intend to delve into particulars of his case. But I became a science writer specifically in order to write about sudden cardiac death in athletes — it was my journalistic raison d'être for years — and I think it’s worth sharing a lesson or two from my past reporting, and perhaps a tip on what all of us can do to save some lives.
If you just want the tips, you can skip to the last section. Otherwise…
A little person background
The pictures below are a few I still have from high school, showing me and my 4x800-meter relay teammates, who also happened to be my closest friends.



In the picture of four guys facing the camera, I’m the short one. In the other two – one warming up, one mid-race — I think you can find me. In the race photo, my teammate, Kevin Richards, is in third. He’s the second-shortest guy in the posed photo.
Two years after that photo was taken, Kevin finished a close second to one of the better milers in the country, took a few steps to recover, and fell to the ground in cardiac arrest. He died there on the indoor track at our high school. My desire to understand what happened ultimately led me to investigate Kevin’s death. His mother signed a waiver allowing me to gather up his medical records. I wound up determined to share what I learned with the world, and decided to try to make my way to Sports Illustrated specifically with the goal of writing about sudden cardiac death in athletes. Which I did, when one of the top-ranked marathoners in the country dropped dead near our Manhattan office during the Olympic Trials in 2007, seven years after Kevin’s death.
Kevin died of hypertrophic cardiomyopathy, or HCM. HCM is usually described as an enlargement of the heart, which is true, but specifically the enlargement is in the wall of the heart’s left ventricle — the chamber that takes oxygenated blood from the lungs and sends it hurtling through the body. And it’s not just that; the cells of that wall, rather than being stacked flush like bricks in a wall, are all askew, like bricks tossed in a dumpster. An electrical signal crossing them and cueing the heart to beat can travel an errant path and cause a fatal misfire. HCM is the condition I usually focused on in my writing about death in athletes, both because it killed my friend, and because it is sometimes cited as the most common cause of sudden cardiac death in people under 35. About 1 in 500 people have it, but few will drop dead, and many will never even know.
Kevin’s death, in time, also ignited my interest in genetics.
What HCM Taught Me About Genetics
HCM, unlike most diseases from which we suffer, is caused by a single genetic mutation. That is: the defect in Kevin’s heart was caused by something akin to a single typo in a dozen complete sets of the Encyclopedia Britannica.
When I first learned that, it felt devastating. As if the minuteness of this error meant that we should have caught and corrected it somehow. Of course, that is completely illogical. And in another way, I found it heartening: it should be easy to screen Kevin’s family members, and people in general, for a single gene mutation.
I dove into the scientific literature, and learned that in the 1990s researchers discovered a specific gene mutation that caused HCM. Hooray! But then they found another. And another. And then hundreds more, and counting. The more families with HCM that have been studied, the more mutations have been found. And to make matters more complicated, most of the HCM-causing gene mutations that have been discovered have been found in only a single family — so-called “private” mutations. Those aren’t useful for widespread testing.
On the bright side, though, a small number of HCM-causing mutations are much more common than others, so a family could be tested for those common mutations; if there’s a hit, they have HCM in the family. If there isn’t, though, there still may be HCM in the family. So you could have hundreds of families (and probably more), all with the exact same disease, none of which have the same genetic mutation.
Thus, the genetic screening situation is actually pretty complicated, and that’s for a very simple disease, in the sense that it is caused by a single mutation. In learning about HCM, I began to learn how really complicated genetic research is, and why 20 years of promises about genetically personalized medicine have failed to materialize. Even the “simple” diseases aren’t simple. And for more common health problems, the situation is much more difficult. Most common illnesses — like most traits in general — are probably influenced by many, many, many parts of an individual’s genome. Each of those interacting parts alone only has a tiny effect, often too tiny to detect. That makes it very hard to study at the genetic level. And that’s aside from the complicated stew of interactions between genes and environmental influences. If you want a deeper dive on why genetically personalized medicine mostly never arrived, here’s a nice (and short) academic explainer by Michael Joyner (Mayo Clinic) and Nigel Paneth (Michigan St.).
If I’d created a motto while working on my first book, The Sports Gene, it could’ve been: “Genetics: more complicated than we thought.” (Borrowed from Laura Hercher.) And on a personal level I learned that genetic screening for common mutations made sense in a family with suspected or confirmed HCM — like my friend Kevin’s family — but that the idea of genetically screening every athlete for HCM in the hope of eradicating sudden death wasn’t sensible.
ECG screening then!
Nobody ever really suggested genetically screening every athlete anyway. What activists, a group of doctors, and communities that had lost someone often have advocated for is universal screening using an ECG, or electrocardiogram, which records the heart’s electrical activity.
Sometimes, in the wake of the death of a high school athlete, a community will raise money and get ECGs for every athlete. I attended a few such screening programs to see how they worked. The programs were absolutely heartwarming, but, I think, not a great solution.
Doctors and activists I spoke to who supported those programs often pointed to Italy, where all athletes — from elementary school to pros — undergo government-subsidized screening. If Italy could do it, why can’t we?
Well, Italy basically had open admissions to med school after World War II. The U.S. has about 2.5 doctors per thousand people; Italy has 8. (I confess that seems a bit high to me, and there may be a discrepancy in data from the WHO and the World Bank. But even the lower figures put Italy at 4.) Many Italian physicians have spent their entire careers screening athletes with ECGs. The U.S. has fewer doctors per capita, a far more genetically and geographically diverse population, and way more high school athletes than Italy has teenagers.
As Lisa Salberg, founder of the Hypertrophic Cardiomyopathy Association, once told me: "The U.S. healthcare system does not have the mechanisms to pay for an ECG for every athlete. Nor do we have enough trained professionals to evaluate the results. There would be a lot of athletes placed on alert for no reason, and a lot of missed diagnoses." (In the years since we discussed it, though, false-positive rates and cost may have declined. And here’s an argument for ECG screening in certain, higher risk athlete populations.)
But she also told me something else:
"Somewhere between screening every kid in America for a couple of billion dollars and not doing anything lies the right answer."
Somewhere between….
When I first got curious about sudden cardiac death and attended a conference, I met people who weren’t quite sure if something deadly ran in their family. Maybe one family lost cousin Jimmy when he drowned in a pool (even though he was a varsity swimmer), and uncle Fred died in a one-car accident. Were those actually sudden cardiac arrest? Sometimes, they would learn, the answer was ‘yes.’
We don’t yet know what caused Damar Hamlin’s collapse, but I saw plenty of speculation on social media about what it might have been. Commotio cordis — a very rare disruption of heart rhythm caused by a blow to the chest — was one popular theory, because Hamlin had just taken a helmet to the chest while making a tackle. Another cause proffered was myocarditis, damage to the heart that can be caused by a virus, and that killed Celtics star Reggie Lewis. Neither of those are genetically inherited.
But conditions like HCM, or long QT syndrome — another that causes sudden death on the field — are passed along a family tree in simple fashion. An HCM mutation follows what is known as an “autosomal dominant” inheritance pattern: it has a 50-50 chance of passing from parent to child. And since the only thing more bitter than losing one family member is losing more than one family member, I came away from my reporting in this area with a few recommendations:
-Everybody should know their family medical history better than they probably do. And preparticipation screening for high school sports could actually be a great time to help with that. The American Heart Association has a simple, 12-step screening process, much of which is just gathering some personal and family history. Do you ever nearly faint? Do you ever have chest discomfort while exercising? (Symptoms of dangerous heart conditions in young people are sometimes mistaken for asthma.) Has anyone in your family died of cardiovascular causes before the age of 50?
This is simple stuff, but when I met families affected by HCM, or long QT, it was often only after multiple deaths in the family that they started compiling a detailed family history and getting to the bottom of it.
-Get CPR certified, and learn to use an automated external defibrillator (AED). You can learn compression-only CPR, i.e. no need for mouth to mouth. And AEDs, which shock the heart back into a normal rhythm, are really easy to use, but it’s still good to be familiar with them before you actually have to use one. I let my certifications lapse in both areas, so just signed up for a Red Cross class next week to get them updated. You can find classes near you, or take a virtual class. Some local governments also offer nearly-free courses.
-Some states allow healthcare practitioners who don’t have any cardiovascular training to do preparticipation screening for high school athletes. If we’re going to do that screening, it should probably be done by someone who has a chance of picking up on a suspicious heart murmur. Find out the situation in your state, and if it isn’t satisfactory and you’re interested in this issue, push for change. (Parent and athlete advocacy matters.)
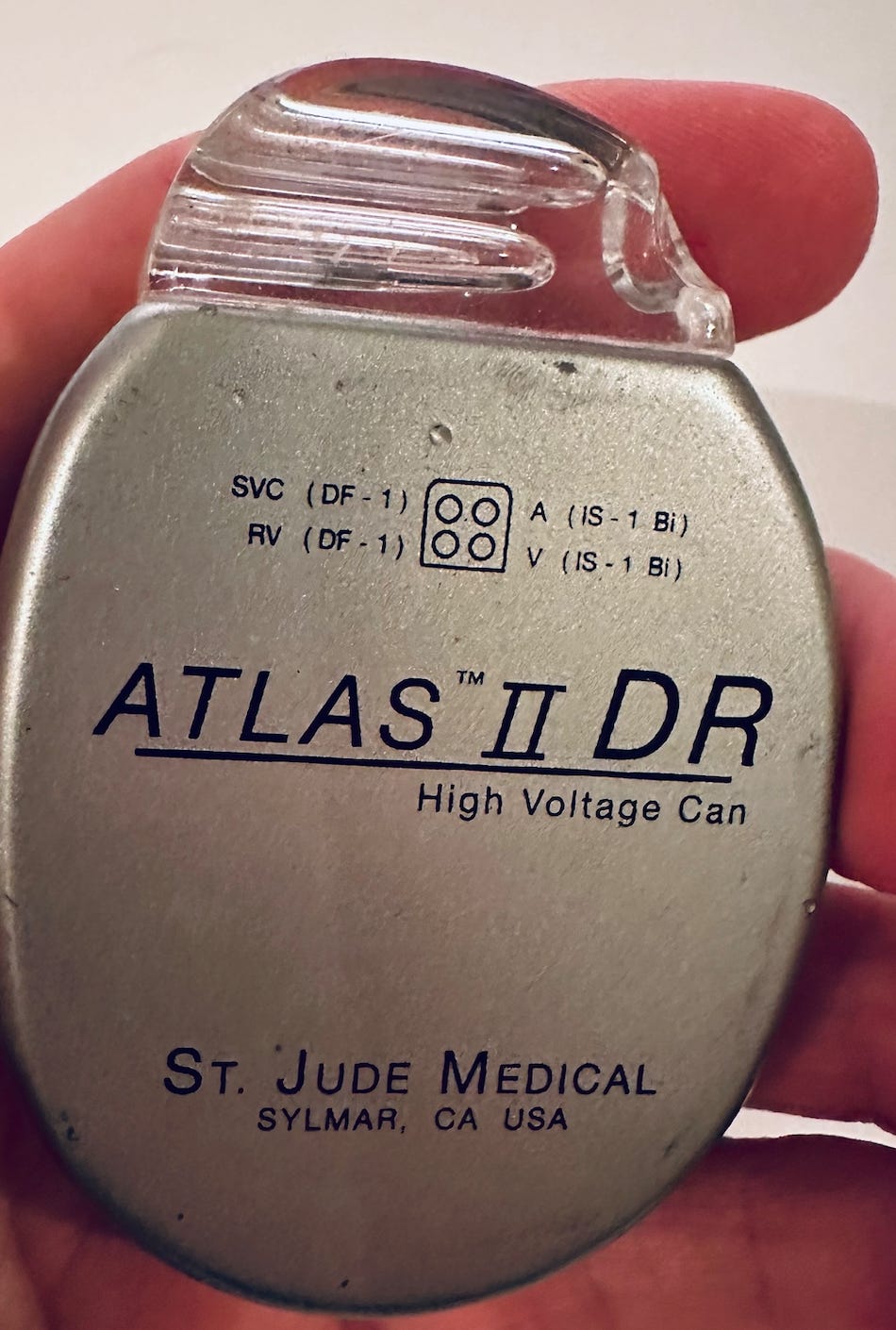
-Finally, if an athlete is confirmed to have HCM, or some other condition that can cause cardiac arrest, they can get one of these little things (pictured below in my hand) implanted in their chest.
That’s an implantable cardioverter defibrillator (ICD). It stands sentinel inside the chest, and if it detects an abnormal heart rhythm it delivers about a 750-volt shock. Not pleasant, but it’ll save a life. In this video, you can see soccer pro Anthony Van Loo collapse on the field, and then a few seconds later he jerks when the ICD shocks him, and then he sits up. Amazing.
In the past, some athletes avoided ICDs because the standard guideline was that, once they get it, they should stop all high-intensity sports, and even overhead weightlifting that might damage wires (known as “leads”) that run from the ICD into the heart. One electrophysiologist I spoke with years ago told me that one of his patients destroyed an ICD just doing aerobics.
But athletes being athletes, some of them have gotten an ICD, and then gone right back to sports anyway. Rachel Lampert, a cardiology professor at Yale, took the opportunity to track them. It has been more than a decade since I first talked to her about this, and at the time her work was still building. So I just searched to see if she had any new and relevant papers, and here one is: a paper she published in 2019 followed 440 athletes with ICDs for up to four years, during which time the ICDs worked beautifully. Sometimes they misfired, shocking someone unnecessarily, but “there were no failures to defibrillate and no injuries related to arrhythmia or shock during sports.”
That is fantastic, and, I must say, a better outcome than I expected years ago. (Sometimes I update my point of view in real time in this newsletter.) Obviously, the decision of whether someone with a potentially fatal cardiac condition should play sports is an individual one that they make with their family and their doctors, but it’s important to know that for those who choose to participate, an ICD can still be a lifesaver.
Thanks so much for reading about an issue that’s very important to me. If you want to read the story of my friend in more detail, check out chapter 15 (“The Heartbreak Gene”) of my book, The Sports Gene.
If a friend sent you this post, you can support Range Widely with a free or paid subscription.
Another way to support this newsletter is to share the post.
Until next time…
David
P.S. I mentioned some of the arguments against universal ECG screening. For those interested in more on the screening debate, here’s a free paper arguing for the merits of preparticipation ECG screening of certain athlete populations.






Here's an interesting discussion between cardiologists on Twitter about whether Hamlin would need an ICD if he's able to (and wants to) return to play:
https://twitter.com/DavidLBrownMD/status/1611134709815447552
This is really interesting. After seeing all of the sudden cardiac arrests on the pitches in the UK, we got to thinking about the grassroots footballers, and what happens to them. 36% of adults in the UK don't know how to perform CPR, so we created a product called the Extra Time Badge: https://www.extratimebadge.com/
In essence, it's a badge that's meant to be ironed on to a uniform / training top in the right place to perform CPR. If that athlete then ever suffers from cardiac arrest, someone close to them can put their hands on the badge and follow the instructions there to perform CPR.
Check it out. It's obviously not going to solve the stuff you wrote above, but it's a first small step.
I'm really sorry to hear about your loss and personal connection to this as well <3